- * 프린트는 Chrome에 최적화 되어있습니다. print
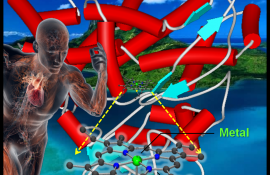
Our research program focuses on biomimetics in small molecule activation by using transition metal model complexes to understand biological systems and design emerging materials. The research requires the tools of chemical synthesis, physicochemical characterization, such as UV-vis, ESI-MS, rRaman, EPR, NMR and X-ray crystallography, and reactivity. It is a multidisciplinary field involving chemistry, biology, medicine, environmental science and others. The current goal of our research is divided into two categories: 1) drug development for anti-tumor activity (e.g., anti-cancer drug) and/or brain plasticity enhancement (e.g., NO delivery); 2) green catalyst synthesis for sustainable environment (e.g., CO2 fixation) and energy conversion (e.g., C1 refinery).
Major research field
Biomimetic Chemistry, Inorganic Chemistry, Coordination Chemistry
Desired field of research
Drug Development, Environmental Catalyst, Energy Conversion
Research Keywords and Topics
The goal of Biomimetic Inorganic-Organic Coordination Chemistry (BIOCC) Laboratory is to clarify the 'Biological Process' in the reactions of metalloenzymes through the biomimetic system and utilize the biomimetic techniques in medical and environmental applications.
# Design of model complexes of metalloenzymes
# Characterization and reactivity studies for biomimetic materials
# Application for medical (e.g., anti-cancer drug, vasodilation) & environmental materials (e.g., catalyst)
Research Publications
D. Jeong, T. Ohta, J. Cho, “Structure and reactivity of a mononuclear nonheme manganese(III)-iodosylarene complex” J. Am. Chem. Soc. 140, 16037, 2018.
D. Jeong, J. J. Yan, H. Noh, B. Hedman, K. O. Hodgson, E. I. Solomon, J. Cho, “Oxidation of naphthalene with a manganese(IV) bis(hydroxo) complex in the presence of acid” Angew. Chem. Int. Ed. 57, 7764, 2018.
H. Noh, D. Jeong, T. Ohta, T. Ogura, J. S. Valentine, J. Cho, “Distinct reactivity of a mononuclear peroxocobalt(III) species toward activation of nitriles” J. Am. Chem. Soc. 139, 10960, 2017.
Patents
J. Cho, D. Seo, J. Choe, Y. Park, S. Shin “Photocleavable cobalt-nitrosyl complex and nitric oxide carrier containing the same” Korea Patent Application Number 10-2019-0103327 (22 Aug 2019)
J. Cho, H. Noh “A method of converting a nitrile functional group into a hydroxamic functional group by using a peroxocobalt complex at room temperature and normal pressure” Korea Patent Application Number 10-2017-0130178 (11 Oct 2017); US Patent Number US 10399928 B2 (3 Sep 2019)
국가과학기술표준분류
- NC. 화학
- NC03. 무기화학
- NC0304. 생무기화학
국가기술지도분류
- 환경/에너지 프론티어 진흥
- 031300. 에너지 소재기술
녹색기술분류
- 에너지원기술
- 재생에너지
- 222. 바이오에너지 기타 기술
6T분류
- BT 분야
- 기초/기반기술
- 020114. 생명현상 및 기능연구




